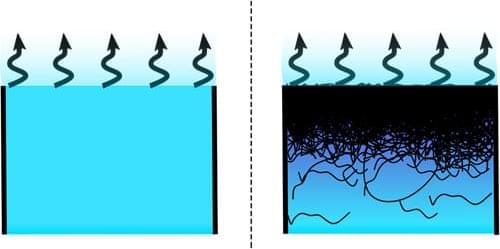
Experiments verify a theory that explains why paint doesn’t dry any faster on a dry day than on a wet day.
You might think that polymer solutions like paint dry more slowly on a humid day than on a dry day. But researchers have now verified a theory that explains why the evaporation rate of the water or another solvent in a polymer solution can be independent of the ambient humidity [1]. The experiments show that, as predicted, water evaporation drives the polymer molecules toward the surface, where they form a dense layer that hinders evaporation and shields the surface from humidity effects. This phenomenon may affect the rate at which virus-containing respiratory droplets evaporate and thus could help explain the seasonal dependence of viral infections.
Humidity-independent evaporation is an advantage in many situations. For example, to preserve the body’s hydration, human skin maintains a nearly constant evaporation rate thanks to cell membranes whose lipid molecules can be reconfigured to adjust the sweat evaporation rate. This reconfiguration is an example of an active process. In 2017, Jean-Baptiste Salmon, a chemical engineer at the University of Bordeaux in France, proposed that humidity-independent evaporation does not require an active response [2]. Instead, his theory suggested that it occurs whenever the solvent evaporates from a solution of large molecules, a process that was already known to draw those molecules toward the drying interface. He predicted that, after the large molecules form a dense layer, the solvent’s evaporation rate will remain unchanged whether the surroundings are bone dry or at 100% humidity. However, the theory has not been tested with a nonactive polymer solution.