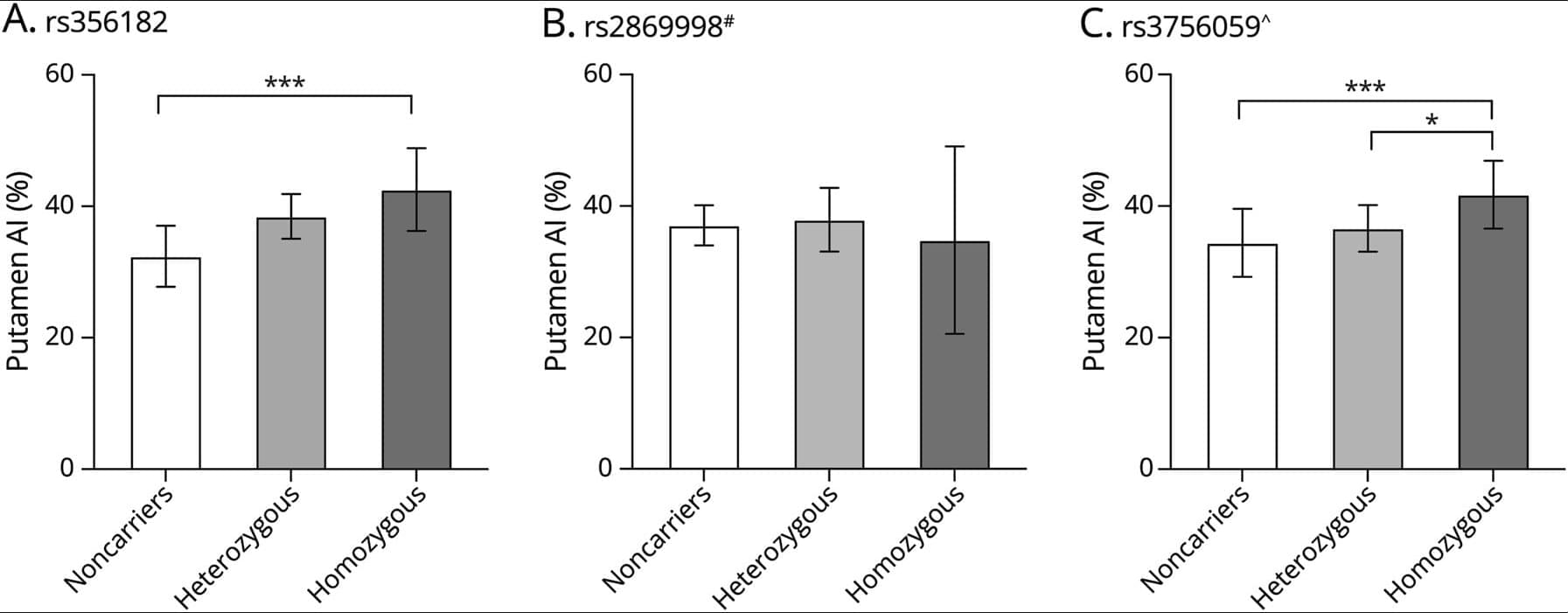
This study assessed the endophenotypic potential of striatal dopamine transporter uptake in carriers of Parkinson disease–associated SNCA genetic risk variants.
ObjectivesThe aim of this study was to investigate the endophenotypic potential of striatal dopamine transporter (DAT) uptake in carriers of Parkinson disease (PD)–associated SNCA genetic risk variants. MethodsWe analyzed 381 patients with de novo PD from the Parkinson’s Progression Markers Initiative (PPMI). The genotype of previously identified PD-related SNCA risk variants was extracted and used to compute an individual PD-specific SNCA genetic risk score (GRS). Striatal DAT uptake was quantified using 123 I‐FP‐CIT SPECT and assessed at baseline and 24-month follow-up. Mixed models were applied to explore the relationship between striatal 123 I‐FP‐CIT SPECT specific binding ratios (SBRs) and PD SNCA risk variants.