Recent studies suggest that the steady rise in life expectancy observed over the past 200 years has now stagnated. Data indicate that a limit has been reached, and that medical and healthcare advances no longer affect longevity in developed countries as they did in previous decades. Today, ageing itself, rather than disease, is the real frontier of human longevity. But what exactly is ageing? And can it be addressed in the same way as a disease?
A research team has just published the final peer-reviewed data from the study of the longest-lived person ever recorded, who far exceeded 117 years: the Catalan woman Maria Branyas. The analysis, based on samples obtained using minimally invasive techniques, takes a multi-omic approach with genomic, proteomic, epigenomic, metabolomic and microbiomic technologies, and represents the most exhaustive study ever undertaken on a supercentenarian.
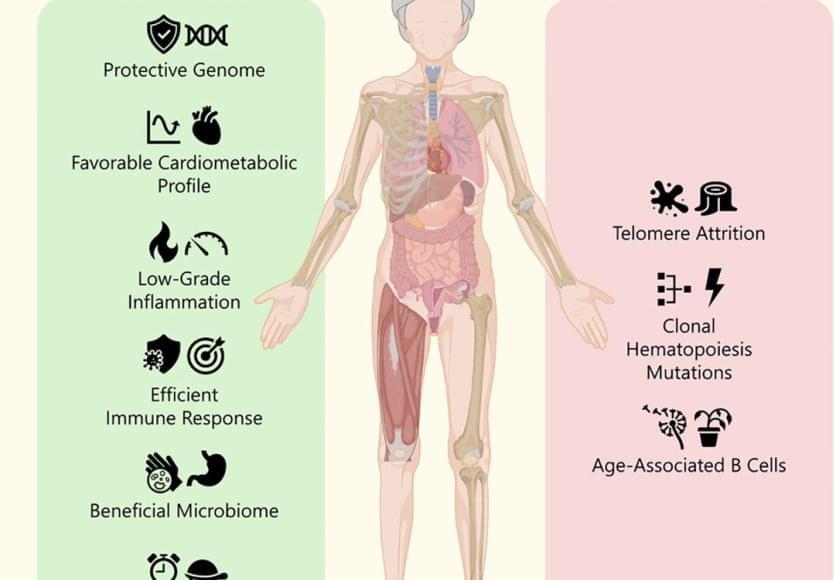
In the paper, published in the prestigious journal Cell Reports Medicine, the international and multidisciplinary team explains that individuals who reach supercentenarian age do not do so through a general delay in ageing but, as the author notes, thanks to a “fascinating duality: the simultaneous presence of signals of extreme ageing and of healthy longevity.”